What Are Intramolecular & Intermolecular Forces? (Answers)
INTRAMOLECULAR & INTERMOLECULAR FORCES – In this article, we will learn about what Intramolecular & Intermolecular forces are.
Intermolecular forces are the one which occurs because of the interaction of one molecule with another that belongs to the same compound.
Intramolecular forces are forces that hold atoms together within a molecule. There are also different types of intramolecular forces of attraction.
Ionic Bond – This bond is formed by the complete transfer of valance electron(s) between atoms. It is also a type of chemical bond that generates two oppositely charged ions.
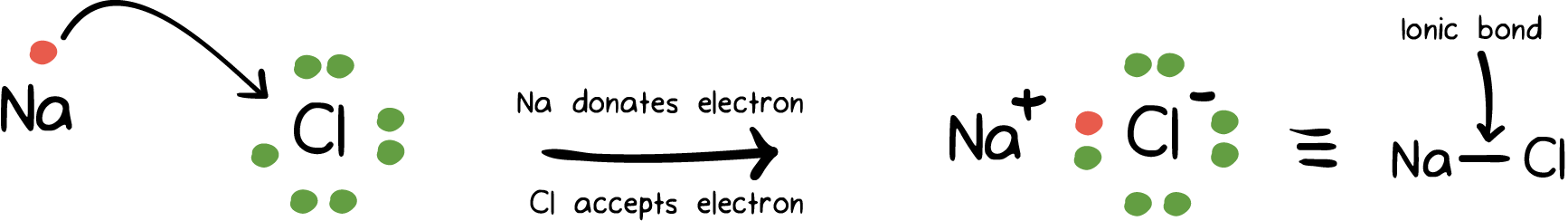
In this type of bond, the metal loses electrons and turns into positively charged cations. However, the non-metal accept those electrons to become negatively charged ones.
Covalent Bond – This is created between atoms that have the same affinity or desire for electrons.
According to an article from Khan Academy, these are called electronegativities. Because both atoms have the same affinity for electrons, neither have a tendency to give them away.

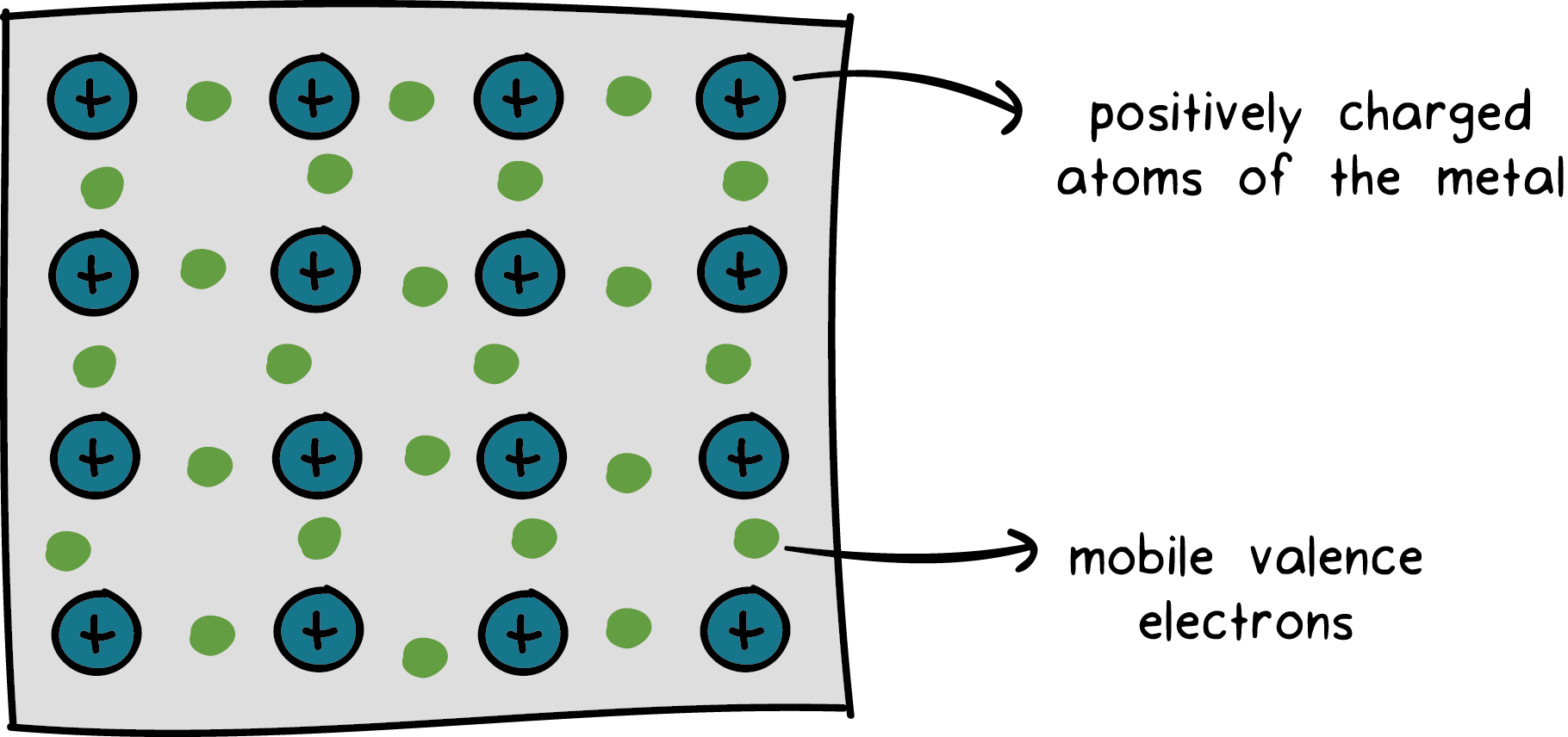
Metallic Bonding – This type of covalent bonding happens between atoms of metal. Here, valence electrons are free to move through the lattice.
This bond is created through the attraction of mobile electrons which is called the sea of electrons.
Here are the types of Intermolecular forces that exist between molecules.
Dipole-dipole interactions – This occurs when the partially positively charged areas of a molecule interacts with the partially negative charged part of the neighboring molecule.
Hydrogen Bonding – This is a special kind of dipole-dipole bonding which occurs between a hydrogen atom bonded to an oxygen, nitrogen, or fluorine atom.
The partially positive end of hydrogen is pulled towards the partially negative end of the oxygen, nitrogen, or fluorine of another molecule.
London dispersion forces – These are the weakest of the intermolecular forces and exists between every type of molecule. The more electrons a molecule has, the stronger this force becomes.

Like this article? READ ALSO: Commonly Asked Science Questions And The Answers To Each
